Water is the most widely used substance, raw material or starting material in the production, processing and formulation of pharmaceutical products. It has unique chemical properties due to its polarity and hydrogen bonds. This means it is able to dissolve, absorb, adsorb or suspend many different compounds. These include contaminants that may represent hazards in themselves or that may be able to react with intended product substances, resulting in hazards to health.
Pharmaceutical Water System Ppt – What Is Pharmaceutical Water
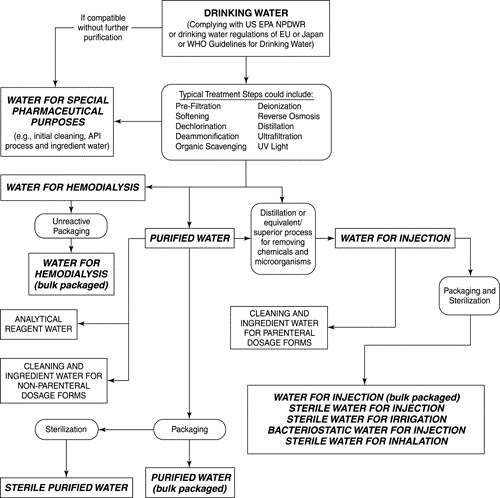
Water is used as ingredient, and solvent in the processing, formulation, and manufacture of pharmaceutical products, active pharmaceutical ingredients (APIs) and intermediates, compendial articles, and analytical reagents. This general information chapter provides additional information about water, its quality attributes that are not included within a water monograph, processing techniques that can be used to improve water quality, and a description of minimum water quality standards that should be considered when selecting a water source.
Pharmaceutical water includes different types of water used in the manufacture of drug products.
THE 8 TYPES OF WATER ARE:
Non-potable
Potable (drinkable) water
USP purified water
USP water for injection (WFI)
USP sterile water for injection
LUSP sterile water for inhalation
USP bacteriostatic water for injection
USP sterile water for irrigation
Control of the chemical purity of these waters is important and is the main purpose of the monographs in this compendium. Unlike other official articles, the bulk water monographs (Purified Water and Water for Injection) also limit how the article can be produced because of the belief that the nature and robustness of the purification process is directly related to the resulting purity. The chemical attributes listed in these monographs should be considered as a set of minimum specifications. More stringent specifications may be needed for some applications to ensure suitability for particular uses. Basic guidance on the appropriate applications of these waters is found in the monographs and is further explained in this chapter.
Control of the microbiological quality of water is important for many of its uses. All packaged forms of water that have monograph standards are required to be sterile because some of their intended uses require this attribute for health and safety reasons. USP has determined that a microbial specification for the bulk monographed waters is inappropriate and has not been included within the monographs for these waters. These waters can be used in a variety of applications, some requiring extreme microbiological control and others requiring none. The needed microbial specification for a given bulk water depends upon its use.
A single specification for this difficult-to-control attribute would unnecessarily burden some water users with irrelevant specifications and testing. However, some applications may require even more careful microbial control to avoid the proliferation of microorganisms ubiquitous to water during the purification, storage, and distribution of this substance. A microbial specification would also be inappropriate when related to the “utility” or continuous supply nature of this raw material. Microbial specifications are typically assessed by test methods that take at least 48 to 72 hours to generate results. Because pharmaceutical waters are generally produced by continuous processes and used in products and manufacturing processes soon after generation, the water is likely to have been used well before definitive test results are available.
Failure to meet a compendial specification would require investigating the impact and making a pass/fail decision on all product lots between the previous sampling’s acceptable test result and a subsequent sampling’s acceptable test result. The technical and logistical problems created by a delay in the result of such an analysis do not eliminate the user’s need for microbial specifications. Therefore, such water systems need to be operated and maintained in a controlled manner that requires that the system be validated to provide assurance of operational stability and that its microbial attributes be quantitatively monitored against established alert and action levels that would provide an early indication of system control.
Pharmaceutical Water System PPT – What Is Pharmaceutical Water – Principles PDF
Important Notes on Pharmaceutical Water Systems
- Control of the quality of water throughout the production, storage and distribution processes, including microbiological and chemical quality, is a major concern. Unlike other product and process ingredients, water is usually drawn from a system on demand, and is not subject to testing and batch or lot release before use. Assurance of quality to meet the on-demand expectation is, therefore, essential. Additionally, certain microbiological tests may require periods of incubation and, therefore, the results are likely to lag behind the water use.
- Control of the microbiological quality of WPU is a high priority. Some types of microorganism may proliferate in water treatment components and in the storage and distribution systems. It is crucial to minimize microbial contamination by proper design of the system, periodic sanitization and by taking appropriate measures to prevent microbial proliferation.
- Different grades of water quality are required depending on the route of administration of the pharmaceutical products. Other sources of guidance about different grades of water can be found in pharmacopoeias and related documents.
Pharmaceutical Water System: Principles For Pharmaceutical Water Systems
- Pharmaceutical water production, storage and distribution systems should be designed, installed, commissioned, qualified and maintained to ensure the reliable production of water of an appropriate quality. It is necessary to validate the water production process to ensure the water generated, stored and distributed is not beyond the designed capacity and meets its specifications.
- The capacity of the system should be designed to meet the average and the peak slow demand of the current operation. If necessary, depending on planned future demands, the system should be designed to permit increases in the capacity or designed to permit modification. All systems, regardless of their size and capacity, should have appropriate recirculation and turnover to assure the system is well controlled chemically and microbiologically.
- The use of the systems following initial validation (installation qualification (IQ), operational qualification (OQ) and performance qualification (PQ)) and after any planned and unplanned maintenance or modification work should be approved by the quality assurance (QA) department using change control documentation.
- Pharmaceutical Water System PPT – What Is Pharmaceutical Water – Principles PDF Doc
- Water sources and treated water should be monitored regularly for chemical, microbiological and, as appropriate, endotoxin contamination. The performance of water purification, storage and distribution systems should also be monitored. Records of the monitoring results, trend analysis and any actions taken should be maintained.
- Where chemical sanitization of the water systems is part of the biocontamination control programme a validated procedure should be followed to ensure that the sanitizing process has been effective and that the sanitizing agent has been effectively removed.