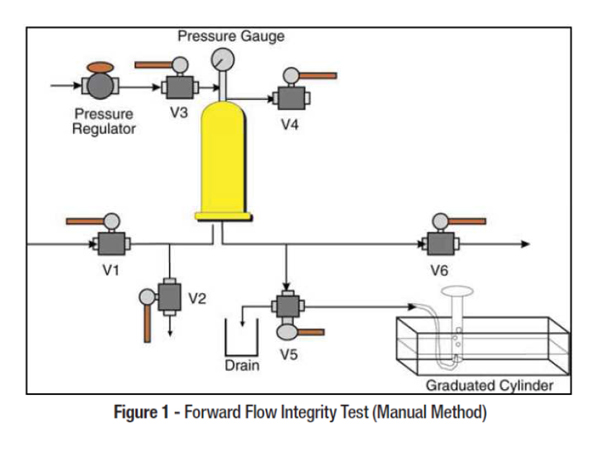
A filter integrity test is a critical unit operation commonly employed in the Pharma industry. FDA Guideline on Sterile Drug Products @ FILTER INTEGRITY TESTING is given below.
FILTER INTEGRITY TESTING
Sterilizing grade filters require testing to assure the filters are integral and fulfill their purpose. Such filter tests are called integrity tests and are performed before and after the filtration process. Sterilizing grade filtration would not be admitted to a process if the filter would not be integrity tested in the course of the process. This fact is also established in several guidelines, recommending the use of integrity testing, pre- and post-filtration. This is not only valid for liquid but also for air filters.
Examples of such guidelines are :
-
FDA Guideline on Sterile Drug Products Produced by Aseptic Processing (1987):
Normally, integrity testing of the filter is performed after the filter unit is assembled and prior to use. More importantly however, such testing should be conducted after the filter is used in order to detect any filter leaks or perforations that may have occurred during filtration.
- The Guide to Inspections of High Purity Water Systems, Guide to Inspections of Lyophilization of Parenterals, and also the CGMP document 212.721 Filters state the following:
- The integrity of all air filters shall be verified upon installation and maintained throughout use. A written testing program adequate to monitor integrity of filters shall be established and followed. Results shall be recorded and maintained as specified in 212.83.
- Solution filters shall be sterilized and installed aseptically. The integrity of solution filters shall be verified by an appropriate test, both prior to any large-volume parenteral solution filtering operation and at the conclusion of such operation before the filters are discarded. If the filter assembly fails the test at the conclusion of the filtering operation, all materials filtered through it during that filtering operation should be rejected. Rejected materials may be refiltered using filters whose integrity has been verified provided that the additional time required for refiltration does not result in a total process time that exceeds the limitations specified in 212.111. Results of each test shall be recorded and maintained as required in 212.188(a).
- ISO 13408-1 First Edition, 1998-08-1, Aseptic Processing of Health Care Products, Part 1: General requirements: Section 17.11.1 Investigation, m. pre- and post-filter integrity test data, and/or filter housing assembly:
- 20.3.1. A validated physical integrity test of a process filter shall be conducted after use without disturbing the filter housing assembly. Filter manufacturer’s testing instructions or recommendations may be used as a basis for a validated method. Physical integrity testing of a process filter should be conducted before use where process conditions permit. ‘‘Diffusive Flow,’’ ‘‘Pressure Hold,’’ and ‘‘Bubble Point’’ are acceptable physical integrity tests.
- 20.3.2. The ability of the filter or housing to maintain integrity in response to sterilization and gas or liquid flow (including pressure surges and flow variations) shall be determined.
- USP 23, 1995, P. 1979. Guide to Good Pharmaceutical manufacturing Practice (Orange
FDA Guide, U.K., 1983):
- PDA (Parenteral Drug Association), Technical Report No. 26, Sterilizing Filtration of Liquids (March 1998):
Integrity tests, such as the diffusive flow, pressure hold, bubble point, or water intrusion tests, are non-destructive tests, which are correlated to the destructive bacteria challenge test with 107/cm2 B. diminuta. Derived from these challenge tests, specific integrity test limits are established, which are described and documented within the filter manufacturers’ literature. The limits are water-based; i.e., the integrity test correlations are performed using water as a wetting medium. If a different wetting fluid, such as a filter or membrane configuration, is used, the integrity test limits may vary. Integrity test measurements depend on the surface area of the filter, the polymer of the membrane, the wetting fluid, the pore size of the membrane, and the gas used to perform the test.
Wetting fluids may have different surface tensions, which can depress or elevate the bubble point pressure. The use of different test gases may elevate the diffusive gas flow. Therefore, appropriate filter validation has to be established to determine the appropriate integrity test limits for the individual process. Bubble Point Test Microporous membranes will fill their pores with wetting fluids by imbibing that fluid in accordance with the laws of capillary rise. The retained fluid can be forced from the filter pores by air pressure applied
from the upstream side. The pressure is increased gradually in increments. At a certain pressure level, liquid will be forced first from the set of largest pores, in keeping with the inverse relationship of the applied air pressure P and the diameter of the pore, d, described in the bubble point equation:
where g is the surface tension of the fluid, y is the wetting angle, P is the upstream pressure at which the largest pore will be freed of liquid, and d is the diameter of the largest pore.
When the wetting fluid is expelled from the largest pore, a bulk gas flow will be detected on the downstream side of the filter system (Fig. 7). The bubble point measurement determines the pore size of the filter membrane, i.e., the larger the pore the lower the bubble point pressure. Therefore, filter manufacturers specify the bubble point limits as the minimum allowable bubble point. During an integrity test, the bubble point test has to exceed the set minimum bubble point.
Manual bubble point test set up
1.Diffusion Test
A completely wetted filter membrane provides a liquid layer across which, when a differential pressure is applied, the diffusive airflow occurs in accordance with Fick’s law of diffusion. This pressure is called test pressure and commonly specified at 80% of the bubble point pressure. In an experimental elucidation of the factors involved in the process, Reti simplified the integrated form of Fick’s law to read as follows:
where N is the permeation rate (moles of gas per unit time), D is the diffusivity of the gas in the liquid, H is the solubility coefficient of the gas, L is the thickness of liquid in the membrane (equal to the membrane thickness if the membrane pores are completely filled
with liquid), P (p1 _ p2) is the differential pressure, and r is the void volume of the membrane, its membrane porosity, commonly around 80%. The size of pores only enter indirectly into the equation; in their combination, they comprise L, the thickness of the liquid layer, the membrane being some 80% porous. The critical measurement of a flaw is the thickness of the liquid layer. Therefore, a flaw or an oversized pore would be measured by the thinning of the liquid layer due to the elevated test pressure on the upstream side. The pore or defect may not be large enough that the bubble point comes into effect, but the
test pressure thins the liquid layer enough to result into an elevated gas flow. Therefore, filter manufacturers specify the diffusive flow integrity test limits as maximum allowable diffusion value. The larger the flaw or a combination of flaw, the higher the diffusive flow.
Pressure Hold Test:
The pressure hold test is a variant of the diffusive airflow test. The test set-up is arranged as in the diffusion test except that when the stipulated applied pressure is reached, the pressure source is valved off. The decay of pressure within the holder is then observed as a function of time, using a precision pressure gauge or pressure transducer.
The decrease in pressure can come from two sources:
1) the diffusive loss across the wetted filter. Because the upstream side pressure in the holder is constant, it decreases progressively as all the while diffusion takes place through the wetted membrane and
2) the source of pressure decay could be a leak of the filter system set-up. An important influence on the measurement of the pressure hold test is the upstream air volume within the filter system. This volume has to be determined first to specify the maximum allowable pressure drop value. The larger the upstream volume, the lower will the pressure drop be. The smaller the upstream volume, the larger the pressure drop. This also means an increase in the sensitivity of the test, and also an increase of temperature influences, if changes occur. Filter manufacturers specify maximum allowable pressure drop values.
2.Water Intrusion Test:
The water intrusion test is used for hydrophobic ventand air membrane filters only. The upstream side of the hydrophobic filter cartridge housing is flooded with water. The water will not flow through the hydrophobic membrane. Air or nitrogen gas pressure is then applied to the upstream side of the filter housing above the water level to a defined test pressure. This is done by way of an automatic integrity tester. A period of pressure stabilization takes place over time frame, by the filter manufacturer’s recommendation, during which the cartridge pleats adjust their positions under imposed pressures.
After the pressure drop thus occasioned stabilizes, the test time starts, and any further pressure drop in the upstream pressurized gas volume, as measured by the automatic tester, signifies a beginning of water intrusion into the largest (hydrophobic) pores, water being incompressible. The automated integrity tester is sensitive enough to detect the pressure drop. This measured pressure drop is converted into a measured intrusion value, which is compared to a set intrusion limit, which has been correlated to the bacteria challenge test. As with the diffusive flow test, filter manufacturers specify a maximum allowable water intrusion value. Above this value, a hydrophobic membrane filter is classified as non-integral.
References for FILTER INTEGRITY TESTING:
- Cooper and Gunn’s. Tutorial Pharmacy by S.J.Carter.
- Pharmaceutical engineering; K. Sambamurthy
- Pharmaceutical engineering; principles and practices, C.V.S. Subrahmanyam
- Encyclopedia of pharmaceutical technology, vol 3, edited by James Swarbrick.
- Pikal, M.J.; Lukes, A.L.; Lang, J.E. Thermal decomposition of amorphous beta-lactam antibacterials. J. Pharm. Sci. 1977, 66, 1312–1316.
- Pikal, M.J.; Lukes, A.L.; Lang, J.E.; Gaines, K. Quantitative crystallinity determinations of beta-lactam antibiotics by solution calorimetry: correlations with stability. J. Pharm. Sci. 1978, 67, 767–773.
Pikal, M.J.; Dellerman, K.M. Stability testing of pharmaceuticals by high-sensitivity isothermal calorimetry at 25_C: cephalosporins in the solid and aqueous solution states. Int. J. Pharm. 1989, 50, 233–252.
FILTER INTEGRITY TESTING PDF – FDA Guideline on Sterile Drug Products PDF
FILTER INTEGRITY TESTING DOC – FDA Guideline on Sterile Drug Products
FILTER INTEGRITY TESTING PPT
FILTER INTEGRITY TESTING – FDA Guideline on Sterile Drug Products is helpful we hope. If you have anything to this please write to us.