Pharmacology Notes
Apoptosis
Apoptosis is the programmed cell death. It is used by organisms to control the number of cells and tissue size. The cells during apoptosis shrink, but no uncontrolled release of cell debris into the environment occurs. The immune system usually “cleans up” the dying cells, and the content is recycled.
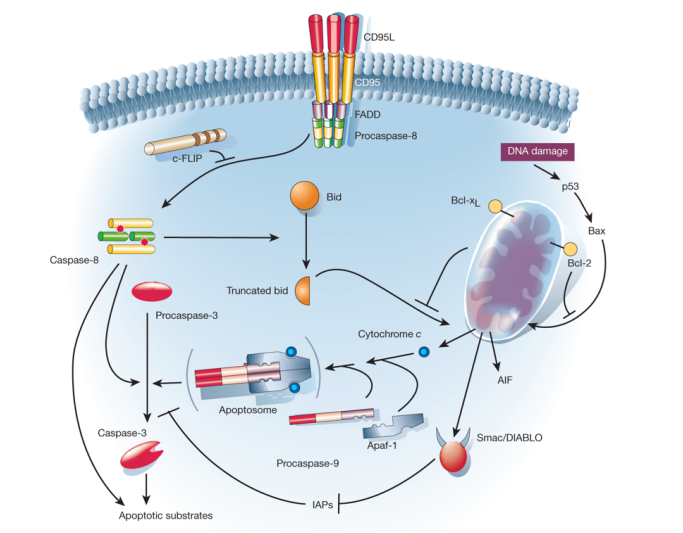
Apoptosis is triggered by an extracellular signal to the CD95 receptor. In response to that signal a set of cysteine proteases, called caspases are activated, that are largely responsible for the morphological changes observed.
Apoptosis Mechanism steps Examples Cancer & Apoptosis PPT PDF
Routes for Apoptosis:
•Two pathways for activation:
i) at the plasma membrane via external ligands upon binding to the death receptor or
ii) via the mitochondrial pathway
•Binding of external ligands such as tumor necrosis factor receptor (TNFα) to Fas receptors of the TNF superfamily induces receptor oligomerization and formation of a death-inducing signaling complex. This complex recruits, via the adaptor molecule FADD (Fas-associated death domain) multiple Pro-caspase-8 molecules, resulting in caspase-8 activation that finally results in caspase-3 activation
•In the mitochondrial pathway release of apoptogenic factors such as cytochrome c, Apaf-1, caspase-9-containing apoptosome complex and inhibitors-of-apoposis proteins trigger caspase-3 activation
• Links between the two pathways exist.
Apoptosis
Regulators of Apoptosis
• The Bcl-2 family of factors regulate caspase activation either negatively ( e.g. Bcl-2, Bcl-XL, MCL1) or positively (e.g. Bcl-XS, Bax,
BAD, BAK, BID)
• The inhibitors of apoptosis proteins (IAP) retard apoptosis
• Upstream modulators are oncogenes such as c-myc, that activates apoptosis in a manner important in cancer therapy • the tumor suppressor p53 induces apoptosis under certain circumstances
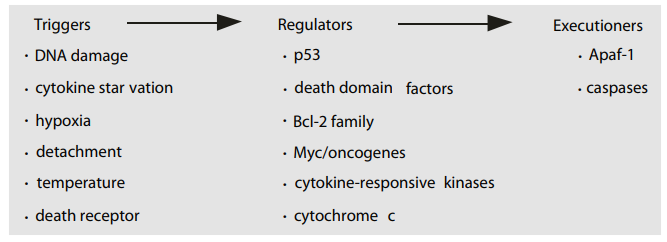
Terms:
Triggers Regulators Executioners
DNA damage
cytokine star vation
hypoxia
detachment
temperature
death receptor
p53
death domain factors
Bcl-2 family
Myc/oncogenes
cytokine-responsive kinases
Apaf-1
caspases
cytochrome c
Incoming Searches
apoptosis examples
apoptosis mechanism
why is apoptosis important
apoptosis steps
apoptosis pathway
apoptosis pdf
apoptosis cancer
apoptosis vs necrosis
Apoptosis vs Necrosis
• Necrosis is the uncontrolled (pathological) cell death. In contrast with apoptosis, cleanup of cell debris by phagocytes of the immune system is generally more difficult. There are many causes of necrosis including injury, infection, cancer, infarction, toxins and inflammation. Necrosis can arise from lack of proper care to a wound site. Usually cell outlines do not stay intact, and cell debris is released into the environment
• Apoptosis is the programmed cell death. It is used by organisms to control the number of cells and tissue size. The cells during apoptosis shrink, but no uncontrolled release of cell debris into the environment occurs. The immune system usually “cleans up” the dying cells, and the content is recycled. Apoptosis is triggered by an extracellular signal to the CD95 receptor. In response to that signal a set of cysteine proteases, called caspases are activated, that are largely responsible for the morphological changes observed.

![Pharmaceutical Water Systems Pharmaceutical Water Storage & Distribution Systems [PDF PPT]](https://pharmawiki.in/wp-content/uploads/2017/11/Pharmaceutical-Water-Systems-Pharmaceutical-Water-Storage-Distribution-Systems-PDF-PPT.jpg)