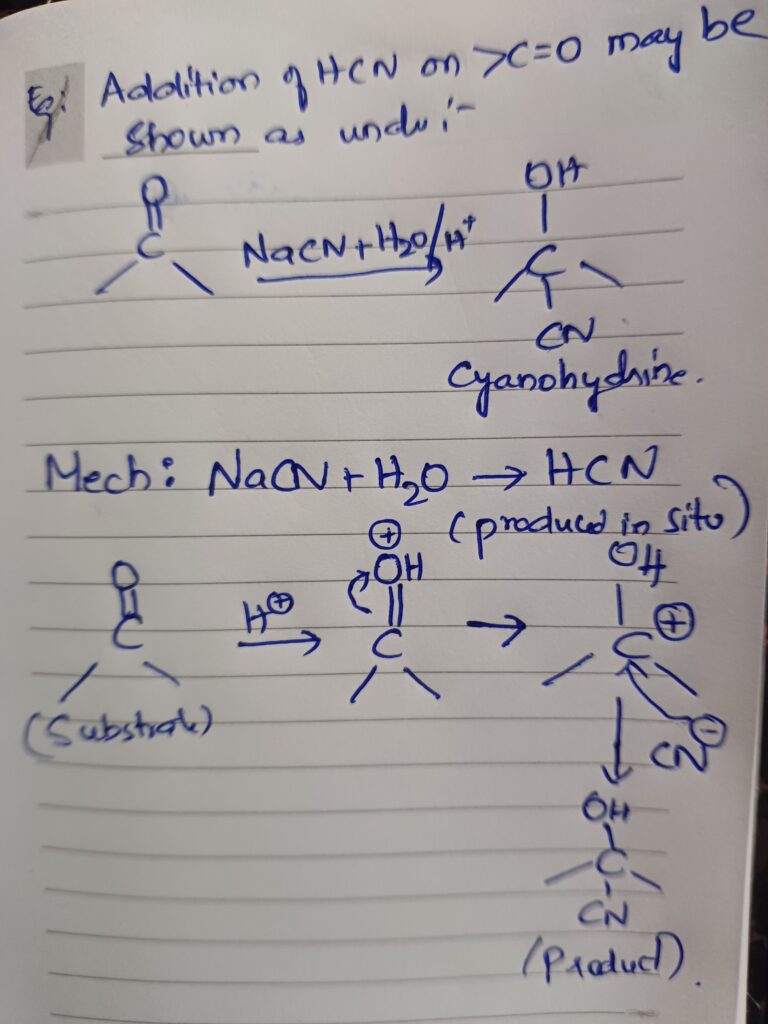
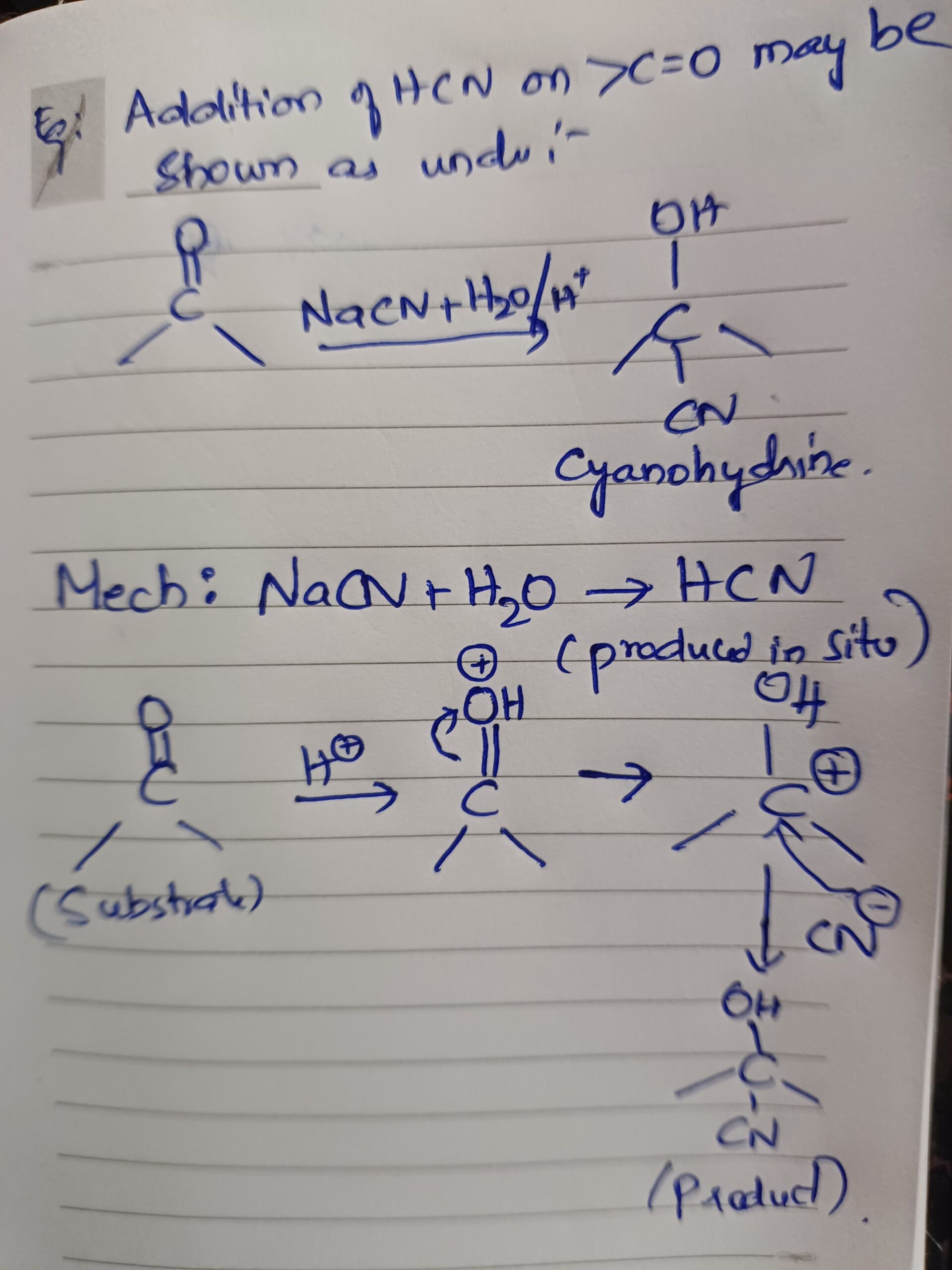
What are Local anaesthetics?
Local anaesthetics – Local anesthetics are drugs which produce insensitivity in a limited area around the site of application or injection of the drug by preventing generation and conduction of impulses along nerve fibers and nerve ending and the effects are reversible.
What are Anthelmintics
Anthelmintics – The drugs which are used to kill or remove the parasitic worms, the term anthelmintic should not be restricted just to drugs acting locally to expel worms from the g.i.t. Various types of worms are able to penetrate tissues, & the drugs used to act against systemic infections should be included also under the general term anthelmintic.
What are anticoagulants?
Anticoagulants – An anticoagulant is a substance that prevents coagulation; that is, it stops blood from clotting & anticoagulants are given to people to stop thrombosis (blood clotting inappropriately in the blood vessels).
What are Diagnostic agents?
Diagnostic agents – These are the agents or chemicals used to detect abnormalities in tissues & organs or to test an organ function, these are thus useful for the clinical diagnosis of the diseases & these agents do not usually have any medicinal values or pharmacological effect.
What are sympathomimetics?
Sympathomimetics – Drugs that mimic the actions obtained as a result of stimulation of the sympathetic or adrenergic nerves are called Sympathomimetics.
OR
The drugs that produce pharmacological effects like adrenaline or nor adrenaline or drugs which bring about stimulation of adrenergic nerves are called Sympathomimetics.
Diuretics – Drugs which promote excretion of water & electrolytes from body through kidneys in the form of urine are called diuretics.
Define antimalarials, Classify them with suitable examples and give the structure of
d)
Pyrimethamine.
Anti-malarial drugs: – The drugs which are used in the treatment of malaria caused due
definition
to Plasmodium Species like Plasmodium Vivax, P. falcifrum, P.malariae, P. ovale are called as Anti-malarial drugs.
Classification:
Quinine salts e.g. Quinine sulphate, Quinine phosphate, Quinine dihydrochloride.
8-Aminoquinolines e.g. Pentaquine, Isopentaquine, Pamaquine, Primaquine.
4-Aminoquinolines e.g. Chloroquine , Amodiaquine.
9-Aminoacridines e.g. Quinacrine, Mepacrine.
Biguanides e.g. Proguanil, Cycloguanil
e.g. pyrimethamine.
Artemisinin & its derivatives.
Miscellaneous: – They are further classified as mentioned below
Sulfones & sulfonamides.
Antibiotics
Write physiological actions of histamine. Classify antihistaminics with examples.
Histamine is a biogenic amine involved in local immune responses as well as regulating physiological function in the gut and acting as a neurotransmitter.
Histamine triggers the inflammatory response. As part of an immune response to foreign pathogens, histamine is produced by basophils and by mast cells found in nearby connective tissues.
marks classification
1mark str.
marks physiological actions,
marks classification
Physiological actions of histamine on various organs:
Blood vessels: Histamine causes dilation of blood vessels
Smooth muscle: It causes contraction of smooth muscle (Contraction of bronchi)
Excretory glands: Histamine has stimulant action on excretory glands. It increases nasal, lachrymal and bronchial secretion.
Acid secretion: Histamine increases acid secretion in stomach which causes peptic ulcer
Oedema: Excess secretion of histamine causes accumulation
of fluid and water in the body.
Allergy: It plays an important role in human allergy and allergic reactions.
Classification of antihistaminics:
H1 blockers or H1 antagonist:
Aminoalkylethers/Ethanolamines e.g. Diphenhydramine, Doxylamine
Ethylenediamine e.g.Mepyramine, Tripelennamine, Pyrilamine
Alkylamines/Propylamines e.g. Pheniramine, Chlorpheniramine, Triprolidine d)Phenothiazine derivatives e.g. Promethazine, Trimeprazine
Piperazine derivatives. e.g Meclizine, Cyclizine, Chlorcyclizine
Dibenzocycloheptenes: Cyproheptadine, Azatadine
Second generation antihistaminics: e.g. Cetrizine, Levocetrizine, Fexofenadine, Terfenadine
H2 Blockers or H2 receptor antagonist e.g. Ranitidine, Cimetidine, Famotidine
An inhibitor of histamine release e.g.Sodium Cromoglycate
Define vitamins. Write the important uses of vit. A, Nicotinic acid and ascorbic acid. 1 mark each.
Vitamins may be defined as potent organic substances which are essential for normal growth and maintenance of life of human and animals, which are not able to synthesize in adequate quantity.
Uses of Niacin or Nicotinic acid-
It is used for preventing vitamin B3 deficiency and related conditions such as pellagra.
Biochemically active form of Nicotinic acid is NAD (Nicotinamide adenine dinucleotide) and its phosphate (NADP). These two coenzymes are required in protein and amino acid metabolism and electron transfer reaction in respiratory chain.
It causes peripheral vasodilation
Large dose of nicotinic acid decreases serum cholesterol level.
Uses of Vitamin A-
It is used for treating vitamin A deficiency.
Prevention and treatment of Night blindness, Xerophthalmia and keratomalacia.
A is important for growth, development and maintenance of immune system.
Some people use vitamin A for improving vision and treating eye disorders including age-related macular degeneration (AMD), glaucoma and
Vitamin A is also used for skin conditions including acne, eczema, psoriasis, cold sores, wounds, burns, sunburn.
Uses of Ascorbic acid-
In general this drug is used for the prevention and treatment of scurvy. This condition is caused by a lack of vitamin C often due to a lack of fresh fruit and vegetables. Symptoms of scurvy include a general feeling of being unwell, tiredness, muscle and joint pain, bleeding into the skin, around bones, into joints and from the gums, and loose teeth.
Ascorbic acid is involved in many redox reactions
(ii) Pethidine
Uses of Pethidine-
Analgesic activity: It is used in the treatment of severe pain like labor pain.
Spasmolytic agent: Pethidine is useful in the treatment of spasm of intestine, urinary bladder
Used as a substitute for morphine for the relief of most types of moderate to severe pains.
Used in combination with chlorpromazine & promethazine to produce narcosis.
It also produces mild euphoria.
h)
Give storage conditions for
Heparin
The aqueous solution is stable for at least 7 years at pH 7 to 8.
It is stored in sealed, sterile container so as to exclude microorganism and moisture.
Cyclopropane
It is stored in metal cylinder designed to hold compressed gases and kept in a cool room free from inflammable material.
The whole cylinder is painted orange. The shoulder should be stenciled with name or symbol “C3H6”. The name or symbol should be clearly stamped on the cylinder valve.
d) Write the difference between general anaesthetics and local anaesthetics. Give the
structure and chemical name of procaine.
2 marks for
Structure of procaine
differences
O
H2N C O C2H5
N
C2H5
Chemical name – 4-amino-(2-diethyl amino ethyl) benzoate or 2-(Diethyl amino)
ethyl-4-amino benzoate.
Distinguish between general anaesthetics and local anaesthetics
GENERAL ANAESTHETICS LOCAL ANAESTHETICS
1. General anaesthetics are the agents which It may be defined as any substance
bring about loss of all modalities of sensation, applied topically or by localized
particularly pain, along with a reversible loss
injection or infiltration to dull or block
of consciousness.
. pain sensation.
2. General anesthesia is induced either by Local anesthesia is induced by topical
inhalation of volatile & gaseous anesthetics application of drugs to skin or mucous
like diethyl ether, halothane or parenteral membrane (surface anesthesia) or by
administration of intravenous anesthetics injection into area subjected to surgical
like thiopentone sodium. operation (infiltration anaesthesia) or
injection into dual membrane of spinal
cord (spinal anesthesia)
3. General anaesthesia is produced before Local Anaesthesia is produced in short
carrying out surgical operation or in surgical procedures & in dentistry.
obstetrics.
4.Care of Vital organs essential Care of Vital organs is not essential
E.g. halothane ,cycloprapane etc. E.g. procaine, lignocaine, benzocaine
Define and classify antihypertensive drugs.
Any agent used for reducing elevated blood pressure is known as antihypertensive agent or hypotensive agent.
Antihypertensive agents can be classified as follows below:-
Centrally acting agents: e.g. α-methyldopa, clonidine
Ganglion blockers : e.g. Pentolinium, Mecamylamine
Adrenergic neuron blockers e.g. Reserpine, Guanethidine
β-adrenergic blockers e.g. Propranalol, Atenolol
α-adrenergic blockers e.g. Prazosin, Tolazoline
Direct-acting vasodilators e.g. Hydralazine, Minoxidil
Calcium channel blockers eg. Verapamil
Angiotensin converting enzyme inhibitors (ACE inhibitors) e.g.Captopril, enalapril maleate.
mark definition
marks classification
3. Attempt any THREE of the following:
Write the general uses of diuretics. Give the structure and brand names of frusemide.
General uses of Diuretics:-
Diuretics are used to treat several conditions in medicine. Following are the conditions where diuretics are used
Hypertension or high blood pressure, Acute left ventricular failure or heart failure
Most types of oedema (renal oedema, oedema of pregnancy) or fluid accumulation
Acute renal failure and treatment of kidney stones
To excrete toxins and toxic metabolites out of the body.
To decreases intraocular pressure in glaucoma.
Treatment of hypercalcemia and hyperkalemia
Frusemide: Lasix, Fru, Frusenex, Tebemid etc.
Structure of frusemide:-
Define and classify NSAIDs.
b)
NSAIDs is an abbreviation for a group of agents called Non Steroidal Anti-inflammatory Drugs.
De
A. Nonselective COX inhibitors (conventional NSAIDs)
Salicylates: Aspirin, Diflunisal
Para Amino Phenol Derivatives- Phenacetin, Paracetamol (Acetaminophen)
Pyrazolone derivatives: Phenylbutazone, Oxyphenbutazone
Indole derivatives: Indomethacin, Sulindac
Propionic acid derivatives: Ibuprofen, Naproxen, Ketoprofen, Flurbiprofen
Anthranilic acid derivatives: Mephenamic acid
Aryl‐acetic acid derivatives: Diclofenac.
Oxicam derivatives: Piroxicam
Pyrrolo‐pyrrole derivative: Ketorolac
Preferential COX‐2 inhibitors: Nimesulide, Meloxicam, Nabumetone
Selective COX‐2 inhibitors: Celecoxib, Rofecoxib, Valdecoxib
Write any one important use of Indigo carmine, Evans blue, Fluorescein Sodium and Congo red.
Uses of Indigo carmine
It is administered intravenously to test renal function (by estimating the rate of excretion in urine) & to locate the uretheral orifices.
In the lab it is used as coloring agents.
Uses of Evans blue
Evans Blue is a di-azo compound used to determine blood volume in humans and animals.
The dye combines firmly with plasma albumin when injected into the blood stream and leaves the circulation very slowly.
Marks classification
1 Mark each
Page 17 of 32
MAHARASHTRA STATE BOARD OF TECHNICAL EDUCATION
(Autonoalium, Placidox, Anaxol, Quietal, Diazewok, Zepose, Microdep
.
What is epilepsy? Classify anticonvulsants and write the structure of
Epilepsy is a disease which arises due to the disorders of control nervous system. This disease is characterized by somewhat more or less frequent recurrence of seizures in
which there occur convulsions or other abnormal body movements, which are accompanied by loss or disturbance in consciousness. Anticonvulsants are classified as:
Barbiturates: – Barbitone sodium, Phenobarbitone, Methyl phenobarbitone.
Hydantoins :- Phenytoin, Mephenytoin
Oxazolidinediones :-Trimethadione, Paramethadione
Succinimides :- Ethosuximide, Phensuximide
Benzodiazepines: Diazepam, Clonazepam, Lorazepam, Nitrazepam
Miscellaneous :- Primidone, Carbamazepine, Valproic acid, Phenacemide, Pregabalin, Gabapentin
Phenobarbitone Structure
Define and classify narcotic analgesic drugs.
Narcotic analgesics are derivatives of opium, semi synthetic or synthetic agents having potent analgesic & narcotic activity and effective for the treatment of severe pain. Classification of Narcotic analgesics
Narcotic analgesic are classified as:-
Morphine and related compounds (Natural alkaloids of opium) e.g. Morphine, Codeine.
Semi-synthetic derivatives of morphine- Heroin, Brown Sugar
Marks Classification 1Mark str.
Mark definition
Marks classify.
Synthetic Agents- Methadone, Pethidine, Dextropropoxyphen hydrochloride
Page
What do you know about sex hormones? Give the uses of Progesterone and Cortisone.
Sex hormones are the hormones which are produced mainly in gonads, ovaries or testes. They influence the development and maintenance of the structures directly and indirectly associated with reproduction. Three main types of sex hormones are
Androgenic or anabolic steroids :-
The androgens are mainly able to maintain the development and maintenance of the secondary male sex characters, thereby increasing virility and libido.
Oestrogens :- Oestrogens influence development and maintenance of secondary female sex characters. They are also essential for maintenance of pregnancy. They also exert anabolic effect on protein metabolism & water retention.
Progestogens.:-
Progestogens are necessary for various changes takes place in uterus & vagina during menstrual cycle, for developing mammary tissue and for maintain pregnancy.
Uses of Progesterone:
It is used as a hormonal replacement therapy in deficiency of progesterone.
It is used in treatment of dysfunctional uterine bleeding.
It is also used along with estrogen in menstrual disorders, premenstrual tension
It is used in treatment of neoplasm of breast and endometriosis.
It has also been incorporated into an intra-uterine device for female contraception.
Treatment of habitual abortion.
Maintenance of pregnancy if it occurs.
Uses of Cortisone:
Anti-inflammatory action: Cortisone is a steroid that prevents the release of substances in the body that cause inflammation.
Cortisone is used to treat many different conditions such as allergic disorders, skin conditions, ulcerative colitis, arthritis, lupus, psoriasis, or breathing disorders.
1 Mark sex hormones,
1.5 Marks to uses of each drug
Subject Title: Pharmaceutical Chemistry-I I Subject Code:
Treatment of rheumatoid arthritis and osteoarthritis
Treatment of lung infection
Treatment of allergic conjunctivitis
It has immune suppressant action hence used in organ transplantation and autoimmune disorder.
Treatment of Addison’s disease.
Write the structure and uses of
Atropine
Atropine Uses:
Atropine has antispasmodic action on smooth muscles, hence used for the treatment of gastric and duodenal ulcers and for the relief of renal and biliary colics.
Useful in symptomatic treatment of Parkinsonism.
It is one of the components of pre-anaesthetic medication, where it is given to reduce salivary and bronchial secretions and to diminish the risk of vagal inhibition of the heart.
It is used by ophthalmologist for its mydriatic effects.
Treatment of hyperhidrosis (Abnormal increased sweating)
1Mark Str.
1Mark use.
Propranolol
Propranolol Uses:
Treatment of various cardiac diseases like Cardiac arrhythmia, Arterial hypertension,
Angina pectoris, congestive heart failure, coronary atherosclerosis, tacycardia
Treatment of Pheochromocytoma (cancer of adrenal glands)
Treatment of glaucoma
5. Attempt any THREE of the following.
Write structure, chemical name uses and brand names of Paracetamol
a)
Structure
Chemical name: p-hydroxy acetanilide OR 4-hydroxy acetanilide OR 4-Acetylaminophenol
Uses:
Antipyretic
Analgesics for relief of pain such as headache, toothache, neuralgia, rheumatism.
Brand names- Tylenol, Calpol, panadol, crocin, metacin, valadol, paldesic, Dolo
Define antiseptics and disinfectants. Classify them with examples
b)
Def: Antiseptic and disinfectants are the chemical agents which are employed to destroy
Pharmaceutical Chemistry
or inhibit the growth of pathogenic microorganism. Antiseptics are applied on living tissues while disinfectants are used on inanimates or non living objects.
CLASSIFICATION
1) Alcohols & Aldehydes
E.g. Ethyl Alcohol, Isopropyl alcohol, Formaldehyde
2) Halogen Compounds.
E.g. Chloramine T, Chorhexidine Acetate, Dibromopropamidine Isothionate.
3) Phenols & Related Compounds
E .g. Phenol, Chlorocresol, Chloroxylenol, Cresol, Hexachlorophene, Thymol.
4) Mercury Compounds.
E.g. , Thiomersal, Mercuric chloride
5) Dyes.
E.g. Proflavine Hemisulphate, Acriflavine, Brilliant Green, Crystal Violet (Gentian Violet), Methylene Blue.
6) Surface Active Agents
E.g. Benzalkonium Chloride, Cetrimide, Cetylpyridinium Chloride, Domiphen Bromide,
7) Miscellaneous Agents.
E.g. Dequalinum Sulphate, Nitrofurazone.
Give structure, chemical name and uses of D.E.C.
Structure
M classification
2 M structure
1 M chemical name
1M uses
Page
Chemical name: 7-Chloro-4-[4’-(diethylamino)-1-methyl butyl] amino quinoline
Dosage forms:
Chlroquine Phosphate Injection
Chlroquine Phosphate Tablets
Chlroquine Syrup
Chroquine Sulphate Injection
Chroquine Sulphate tablet
Brand Names: Cadiquin, Cloquin, Emquin, Lariago, Aralen, Avioclor, Quinross, Resochin, Nivaquine
Attempt any THREE of the following:
What are anti-amoebic agents? Classify them with suitable examples.
Anti-amoebic agents: The drugs which are used in the treatment of amoebic infection caused by Entamoeba histolytica are called as antiamoebic drugs.
Classification of antiamoebic drugs:
Drugs of natural origin: g. emetin
Synthetic drugs:
Quinoline derivative e.g. Chloroquine
Halogenated-8-hydroxyquinoline derivative e.g. quinidochlor, Diiodohydroxyquinoline
Nitro-imidazole derivative e.g. Metronidazole, Tinidazole
rks
M definition
M classification
Page 27 of 32
Antibiotic: e.g. Paramomycin, Tetracycline, chlortetracycline, oxythromycin
Organic arsenicals: e.g. carbarsone
Miscellaneous e.g Diloxanide furoate
Explain the process of blood coagulation. Write the structure and chemical name of
Process of blood coagulation:
Thrombin and several clotting factors present in plasma and calcium ions are involved in the coagulation. Process of blood coagulation can be described as follows.
Whenever there is an injury to a blood vessel, there is formation of rough surface. When blood platelets come in contact with such a rough surface, they are injured.
Due to injury, they release the substance called thromboplastin. In the presence of thromboplastin and calcium in the blood plasma prothrombin is converted into thrombin which helps in conversion of fibrinogen to fibrin.
The fibrin is insoluble and forms threads. The threads of fibrin form a net. In the holes of this net, blood cells are entangled. This mass then contracts to form a blood clot.
i) Indomethacine

Uses:
Anti-inflammatory and analgesic in rheumatoid arthritis
Treatment of spondylitis, osteoarthritis and in gout
Treatment of dysmenorrhea and migraine.
Uses:
To relieve bronchial spasm in acute attacks of asthma.
It is used to increase blood pressure in treatment of hypotension.
Intra venous administration of Adrenaline is used to treat acute circulary collapse or cardiac arrest.
Treatment of allergic disorder.
Treatment of superficial bleeding due to its vasoconstriction effect.
Added to local anesthetic to prolong the duration of effect.
It has mydriatic effect.
Define and classify cholinergic drugs. Write the uses of Acetylcholine Definition:
The agents that mimic the action of acetylcholine or produce the effect of parasympathetic nerve stimulation are called as cholinergic drugs or parasympathomimetic agents.
Classification:
Choline esters: Acetylcholine, Methacholine, Carbachol
Cholinomimetic alkaloids: Muscarine, Pilocarpine, Arecholine
Cholinesterase inhibitors (Indirectly acting)
Reversible Inhibitors- Physostigmine, Neostigmine, Pyridostigmine
Irreversible Inhibitors- Organophosphates (Parathion, Malathion), Insecticides.
Uses of Acetylcholine:
It reduces intraocular pressure in glaucoma
In the relief of atony of gut and urinary bladder