WHAT IS ELECTROMERIC EFFECT?
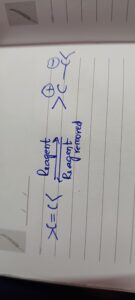
The complete transfer of shared pairs of π electrons of a double bond or multiple bond to one of the bonded atoms under the influence of the attacking reagent is known as electromeric effect.
Is Electromeric effect a temporary effect?
Since the effect involves complete transference of electrons, it lead to the development of full positive charge (+) and negative charges (-) within the molecule. Electromeric effect is a temporary effect and operates in presence of attacking reagent and vanishes as soon as the attacking reagent is withdrawn.
If a proton (H+) adds to C=C bond, the π bond will break and the electron pair will transferred completely to one of the C atoms.
The electromeric effect has no specific direction. Its direction is always that which favours the reaction.
The effect takes place in the direction of the more electronegative element. It can be represented by the symbol E and is of two types: (i) + E effect (ii) – E effect.
What is + E effect and – E effect?
+ E effect: When the displacement of an electron pair is away from the atom or group, it is called + E effect.
-E effect: When the displacement of an electron pair is towards the atom or group, it is called + E effect.
Electromeric Effect explanation with Examples and Mechanism
The Polarizability that is deformation of an unsaturated system on the close approach of a reagent is called electromeric effect. The region when the reagent is removed without allowing the reaction to take place the electronics system reverse to the original ground state of the molecule so this is a temporary effect which operates in the substrate molecule on the demand of the reagent.
This this effect causes complete transfer of those by electrons from one atom to another causing one end positively charged and other negatively charged and the reagent to attack.
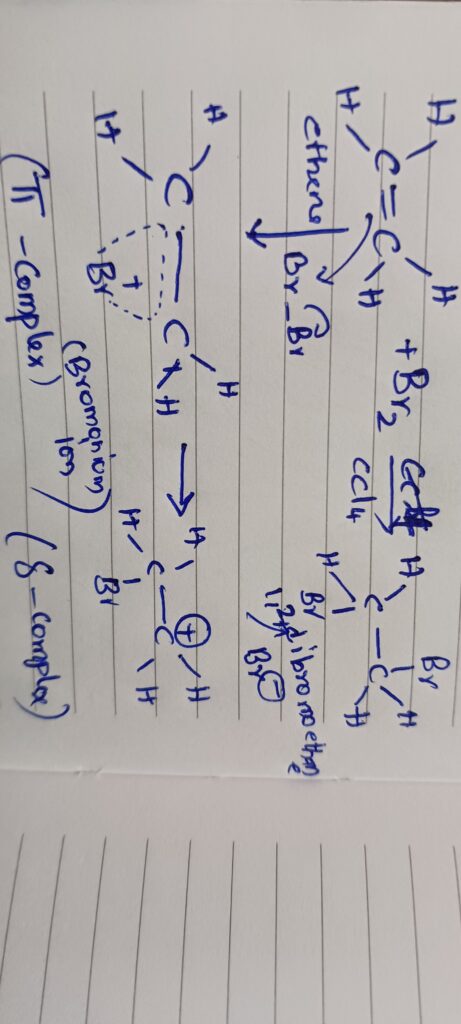
Due to the electromeric effect complete transfer of π electrons take place from one carbon to another through cyclic halonium ion which combines with halide iron to form the product when the multiple bond is between two decimal atoms the shift of electrons takes place towards the more electronegative of the two example edition of reagents on carbonyl compound like aldehydes or ketones.
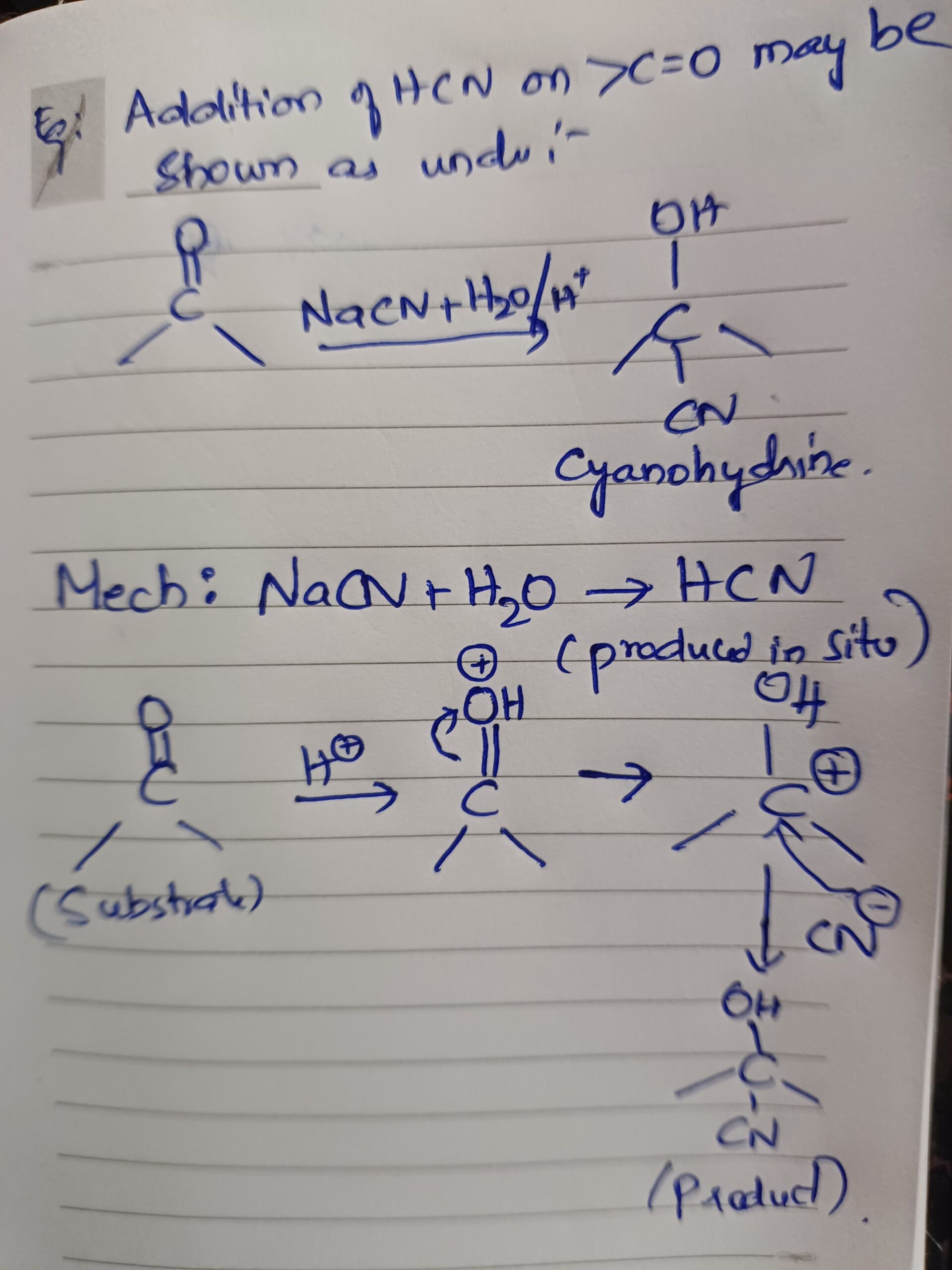
Example edition of hcn on ca=0 maybe shown as under
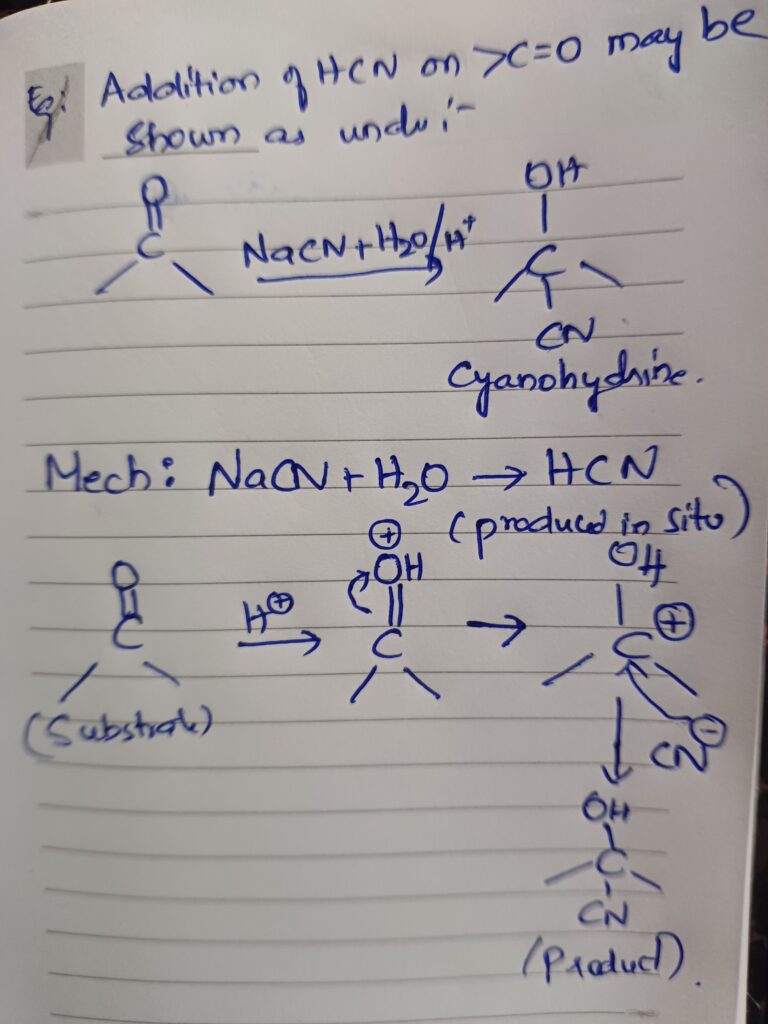
Addition of grignard reagent alcohol NaHSO3 on carbonyl compounds takes place also in this way.
Does the electromeric effect is strong effect than inductive effect since in its effect the loose π electron shift acompletely there by producing full charge + and – on the carbon atoms while in i effect the charge developed on carbon joined to the substituent is small shown as delta §+ § negative.
WHAT IS ELECTROMERIC EFFECT PDF PDF
FOR MORE PHARMACY NOTES please read here.
INDUCTIVE EFFECT
Polarisation of one bond by the influence of an adjacent bond or group is known as Inductive effect. It can be attributed to dipolar interactions through the bonds of a
molecule. Example-The Cl atom in ethyl chloride attracts electron density from C atom to which it is attached and creates a permanent dipole. A carbon chlorine dipole influences
the adjacent C atom by its electron withdrawing inductive effect (-I effect).
Characteristics of Inductive effect:
1. It always causes bond polarization.
2. In C-H, there is almost no inductive effect.
3. Inductive effect is a permanent effect.
4. Inductive influences decreases as the distance from the source increases.